Blueprint for Breakthroughs is a LinkedIn newsletter published by Adrienne B. Mendoza, MHA, SVP BioBridge Global and Chief Operating Officer (COO), BBG Advanced Therapies
Originally published on LinkedIn on March 10, 2026
China’s rise in Advanced cell and gene therapies (CGTs / ATMPs) is less about “competition” and more about system design. China has deliberately built an ecosystem that makes CGT development faster, more scalable, and often more affordable, especially in ex vivo cell therapies—while laying groundwork for in vivo approaches. This article summarizes the key elements of that playbook and what US/EU stakeholders can learn.
1. Where China Is Strong Today: Ex Vivo First, In Vivo Emerging
- China’s current CGT strength is firmly rooted in ex vivo platforms, especially CAR‑T and other T‑cell and stem‑cell therapies run through major academic and hospital centers.
- Hundreds of CAR‑T clinical trials and multiple locally developed CAR‑T products anchor the landscape, making China one of the world’s most active hubs for cell‑therapy research and use.
- In vivo gene therapy and gene‑editing programs are growing on top of this base as regulators introduce specific rules for gene editing and new biomedical technologies, giving sponsors clearer pathways for in vivo trials and future approvals.
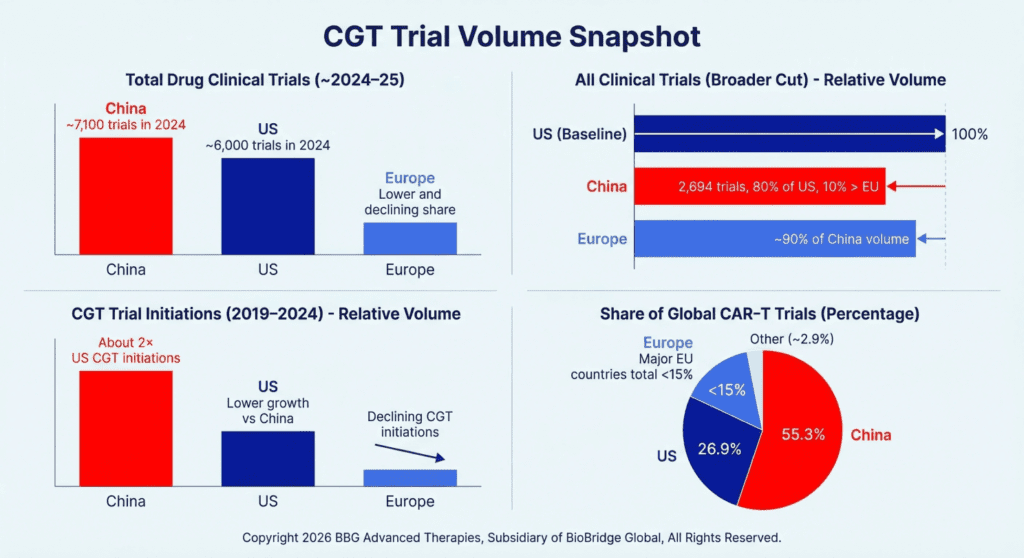
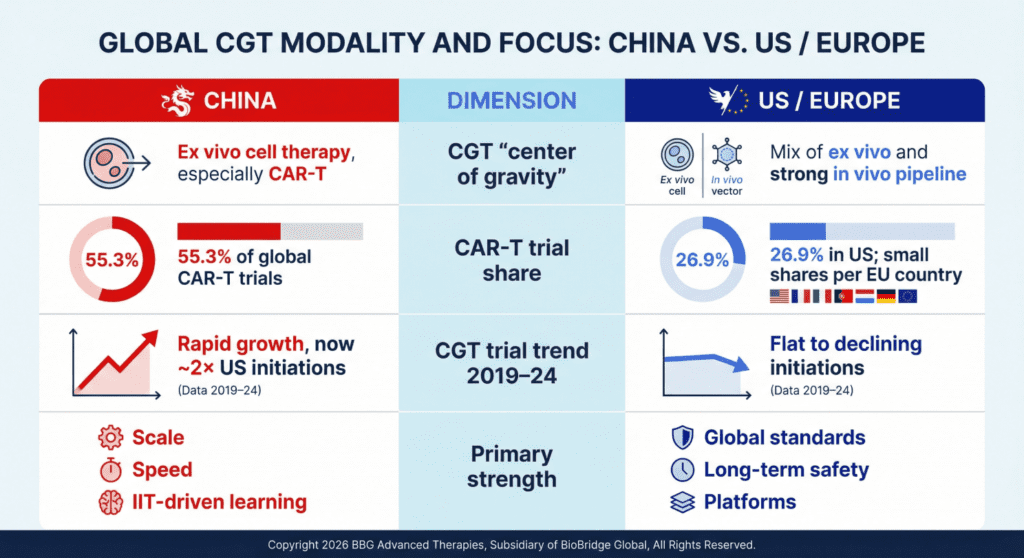
A succinct way to frame it: China is a mature ex vivo engine that is now intentionally building its in vivo capabilities and China has become the largest single country for CGT trial starts, especially in CAR‑T, while US and EU growth has flattened.
2. Regulatory Architecture: Fast Learning, Dual Tracks
China’s regulatory reforms over the last decade have focused on speed and structure for innovative therapies:
- Time‑bounded review (“silent approval”) – Clinical trials move forward if regulators do not object within a set review window, reducing administrative uncertainty.
- Priority and conditional pathways – For serious diseases with unmet need, regulators can grant priority review and conditional approvals tied to ongoing data collection.
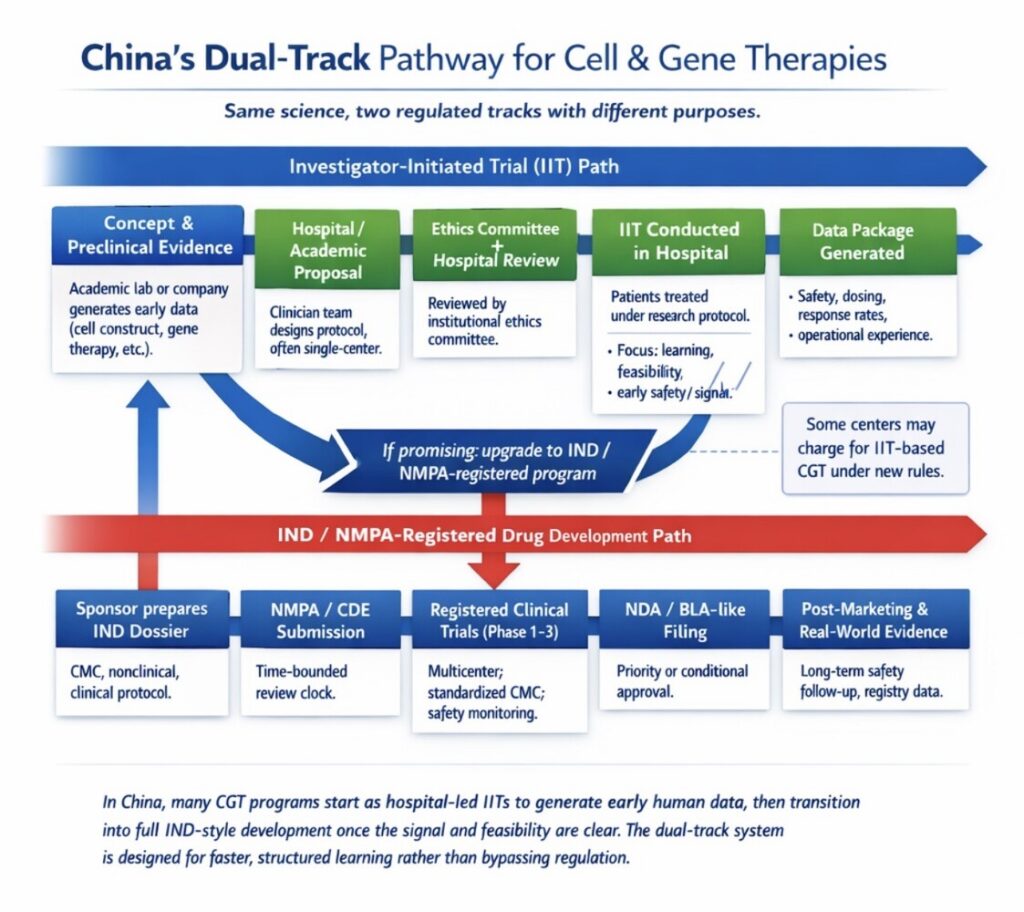
- Dual‑track model (IIT → IND) – A defined distinction exists between: Investigator initiated trials (IITs) and Investigational New Drugs (INDs). Many CGT programs start as hospital‑led IITs to generate early human data, then transition into full IND‑style development once the signal and feasibility are clear. The dual‑track system is designed for faster, structured learning rather than bypassing regulation..
Takeaway for US/EU:
- Preserve high safety standards, but build clearer, faster architectures for early human learning (especially IITs) and for transitions from IIT to formal development.
3. Delivery Model: Community‑Centric and Regional CGT
China’s CGT ecosystem leans heavily on regional centers for both R&D and delivery:
- Major academic hospitals lead numerous CAR‑T and cell‑therapy programs, often designing studies and managing manufacturing workflows.
- Local or regional manufacturing models are common: cells are collected, processed, and reinfused in the same institution or within a nearby network, reducing logistics and shortening vein‑to‑vein times.
- Automated, closed manufacturing platforms are increasingly used to standardize and accelerate production, supporting faster cycles while preserving quality.
This creates a distributed network of CGT “nodes” rather than a small number of centralized factories.
Takeaway for US/EU:
- Treat hospitals and qualified regional centers as potential production nodes for certain CGTs.
- Invest in digital oversight, standardized SOPs, remote QA/QC, and accreditation so decentralization is coupled with robust, consistent quality.
4. Policy, Capital, and Infrastructure: CGT as Strategic Industry
China has explicitly positioned CGT as a strategic sector, aligning policy and investment:
- CGT is embedded in broader innovation and industrial strategies, shaping where public and private capital flows.
- Dedicated guidance for gene and cell therapies clarifies expectations for research, IITs, and clinical use of investigational products.
- Bioparks and innovation zones provide shared infrastructure: lab space, manufacturing shells, testing capacity, and proximity to clinical partners.
- A significant share of domestic biotech financing now targets advanced modalities (cell, gene, RNA), deepening the pipeline and talent base.
Takeaway for US/EU:
- Shift from viewing CGT as a niche to treating it as infrastructure.
- Clarify rules for CGT‑specific issues (IITs, real‑world evidence, post‑marketing demands).
- Build shared backbone assets—apheresis networks, vector capacity, standards and data rails—that individual companies can tap.
5. Access and Affordability: Experiments on Coverage and Cost
China is also experimenting with how to pay for and scale access to CGTs:
- A broad basic medical insurance system covers the majority of the population, providing a base for incremental inclusion of high‑impact therapies.
- CAR‑T prices are generally much lower than typical US list prices, and combined with hospital‑based programs and trials, this can make access more achievable for many patients.
- Selected cities are running policy “sandboxes” for new insurance and long‑term care models, testing how to fund high‑cost, resource‑intensive care.
- Evolving rules around when medical centers may charge for certain investigational therapies (under strict oversight) create a regulated bridge between pure research and full commercialization.
Takeaway for US/EU:
- Use pilots and regional demonstrations to test outcomes‑based, staged, or blended payment models for CGT.
- Integrate CGT planning with disability, chronic care, and long‑term care policies, rather than handling each therapy as a one‑off exception.
- Design ethically robust mechanisms for limited paid access to investigational CGTs when evidence and oversight criteria are met.
6. In Vivo vs Ex Vivo: Strategic Positioning
In simple terms:
- Today: China is primarily an ex vivo cell‑therapy powerhouse (CAR‑T, NK, stem cells) with massive clinical experience and multiple commercial products.
- Emerging: Regulatory updates and new programs are steadily expanding into in vivo gene therapy and gene‑editing approaches, but these are earlier in scale than ex vivo.
This matters for partnering and portfolio strategy: Western companies looking for cell‑therapy partners may find particularly rich opportunities in China’s ex vivo pipeline, while collaborating on in vivo programs will increasingly become important over the coming decade.
7. Big Pharma in China: Commercial and Clinical Footprints
Most major pharma companies now view China as a core part of their CGT strategy:
- AstraZeneca is the clearest “all‑in” example, with long‑standing R&D and manufacturing sites, major new investments earmarked for cell therapy and advanced modalities, and acquisitions to secure end‑to‑end cell‑therapy capabilities.
- Gilead/Kite, BMS, Janssen, Novartis leverage China for CAR‑T and oncology trials, post‑marketing studies, and local collaborations, recognizing the patient pool and trial speed.
- Roche, Sanofi, Pfizer, Merck, GSK, Lilly, Takeda, Bayer and others run large commercial operations and increasingly include Chinese sites in global trials, with growing interest in CGT‑adjacent and CGT‑direct assets.
China is not just a “market” but a discovery, clinical, and BD hub where big pharma sources early‑stage assets and partners with local innovators.
8. From Rivalry to Learning and Partnership
The most productive frame is not “China vs the West” but “complementary strengths”:
- China brings speed, scale, and practical experience in ex vivo cell therapy, hospital‑based delivery, and regulatory experimentation.
- US/EU systems bring deep experience in global quality standards, pharmacovigilance, and long‑term safety, plus strong in vivo and platform‑technology innovation.
For CGT leaders, the key questions are:
- Which elements of China’s regulatory and IIT architecture can inspire more agile, learning‑oriented pathways at home?
- How can hospital‑based manufacturing and regional hubs be adapted to US/EU compliance frameworks?
- Where are the best opportunities for partnering and licensing Chinese CGT assets—especially ex vivo—while contributing global development, manufacturing, and commercialization expertise?
If we treat these as design and collaboration questions rather than a geopolitical scorecard, we have a better chance of delivering safe, timely, and sustainable CGT access for patients everywhere.


