In a recent webinar, Future Proofing Cell & Gene Therapy: Strategies to Secure Your Leukapheresis Supply Chain, Heather Munoz, Principal of Global Market Development, and James Johnson, Director of Starting Materials and Research at BBG Advanced Therapies, discuss today’s most persistent bottlenecks in sourcing well-qualified leukapheresis providers as part of autologous and allogeneic therapy supply chains.
In this article CGT’s next obstacle: Securing the leukapheresis supply chain BBG Advanced Therapies expanding on these topics.
- Leukapheresis as a structural bottleneck
- A much-needed strategic shift: Rethinking vendor strategies for scalable leukapheresis
- Expanding access: Mobile leukapheresis, bridging capacity and access
- Looking ahead
Leukapheresis as a structural bottleneck
Clinical trial activity for CAR-T therapies has surged in recent years. As these therapies expand beyond oncology into larger, chronic indications such as autoimmune and cardiovascular diseases, demand for starting materials is increasing rapidly.
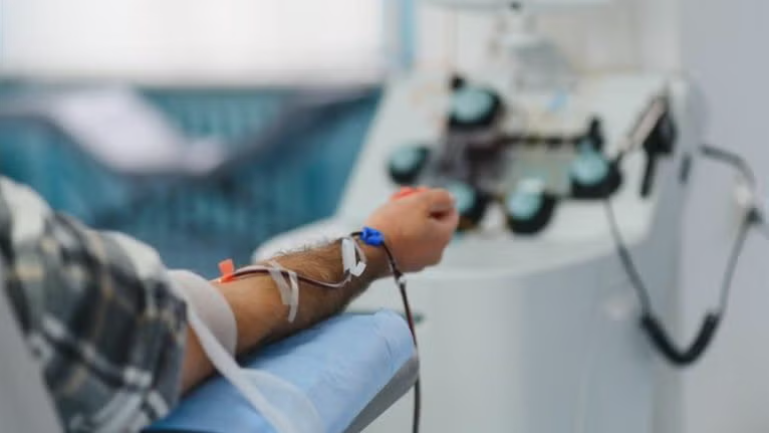
Leukapheresis, the process used to collect immune cells from patients or donors, sits at the very start of this process. It involves circulating blood through a machine to isolate specific white blood cells, with the remaining components returned to the patient over several hours.
While the procedure itself is well established, the current collection model is not built for scale. Most leukapheresis procedures are still concentrated in a limited number of specialised academic medical centres. This creates a structural bottleneck that can delay programme timelines, limit patient access, and introduce variability in starting material quality.
Rethinking vendor strategies for scalable leukapheresis
One of the most forward-looking solutions discussed was the concept of mobile leukapheresis.
BBG Advanced Therapies has developed a mobile leukapheresis center designed to bring collection capabilities closer to patients while maintaining GMP-compliant standards. These mobile units can:
- Provide additional capacity when existing centers are saturated
- Support early-stage and commercial launches
- Expand access to rural and underserved populations
- Improve diversity and representativeness in clinical trials
From a regulatory and operational standpoint, these units are built to mirror traditional facilities, with validated processes, environmental controls, and integrated quality systems. Operating under a centralized quality framework also helps ensure consistency across locations.
Mobile leukapheresis: bridging capacity and access
A highlight of the webinar was a discussion around the BBG Advanced Therapies mobile leukapheresis centre, positioned as a flexible way to support both clinical and commercial demand. Mobile units can provide surge capacity when academic centres are saturated, accelerate early launches while fixed sites come online, and extend access into rural areas, improving trial diversity and real‑world representativeness.
Johnson notes that, from a regulatory perspective, the mobile unit is designed as a like‑for‑like GMP environment with redundant power, environmental monitoring, electronic quality management systems and validated processes stress‑tested in real‑world conditions: “Anything that is necessary in a traditional brick and mortar site is going to be necessary in the mobile setting as well.”
To explore further – including additional detail on regulatory expectations, vendor qualification and chain‑of‑custody controls – download and watch the full webinar here.